Most catalyst research utilizes heated reactors such as fluidized bed reactors (FBR), beginning with multiple parallel trials at the micro-scale, and gradually increasing reactor scale up to the semi-works facility that can study practical catalyst performance at the kilogram scale. Pressures vary from near 1 bar, for environmental catalysis, up to 300 bar for many petrochemical conversions. Reactor pressure control is key to catalyst research.
- Ultra wide flow window, from several liters/minute down to nano flows and zero flow
- Precision typically within 0.5% of set-point in many research applications (5X to 10X more precise than traditional spring regulators)
- Stability during two-phase flow
- High temperature compatibility up to 300C
- compact design easily fits into oven, with set-point controls kept outside oven
- PTFE/Glass diaphragms (most popular for reactor pressure control application)
- Hastelloy C276 and SS316L bodies (typically in stock for quicker response)
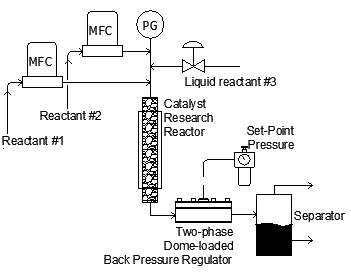
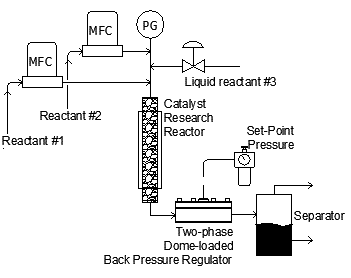
As shown above, mass flow meters are often used to control the input of various gases (and sometimes liquids) into the heated reactor containing the catalyst sample. A back pressure regulator is typically used on the exit of the reactor to maintain stable pressure control for the catalysis process. The products of reaction are monitored using gas chromatography or other forms of analysis.
Application Focus: Fischer-Tropsch Synthesis
Researchers around the world use the Fischer-Tropsch Synthesis process to produce liquid fuels from alternative energy sources such as biomass and natural gas. Fischer-Tropsch Synthesis was developed by Franz Fischer and Hans Tropsch in the early 1900s. It is a relatively complicated chemical process that converts syngas, a mixture of carbon monoxide and hydrogen, into hydrocarbons, including many of the compounds in traditional liquid fuels. Syngas can be generated from almost any carbonaceous feedstock, so the Fischer-Tropsch process indirectly allows for the production of synthetic oil and fuel from resources such as coal, natural gas, and biomass. Equilibar Research Series back pressure regulators are being used in this application. Read the case study on the right for more details.
Application Focus: Two-Phase Flow for Research Pressure Control
Many laboratory catalysis reactors utilize a high pressure liquid separator downstream of the reactor. In addition to requiring an expensive pressure vessel, such processes typically need a liquid level control loop in parallel to the gas phase back pressure regulator. This traditional approach is complex and has several limitations.
Because the Equilibar back pressure valve provides stable pressure control in two-phase flow, it is possible to eliminate the high pressure separator and level control system downstream of the reactor. See how Equilibar back pressure regulators control two-phase flow in the video below.
Contact our engineers to determine which Equilibar back pressure regulator is best suited for your unique two-phase flow application.
Application Focus: Bioreactor Control
A bioreactor supports a biologically active environment, whether it is aerobic or anaerobic. The product is derived from organisms and the bioreactor must therefore be sterile as well as precise in controlling environmental parameters such as temperature, pH and oxygen levels. Equilibar back pressure regulators can be configured with tri-clamp fittings and sterile design features to meet the needs of bioreactor control.

Equilibar® back pressure regulators are ideal to control reactor pressure due to their high precision, wide flow range, and two-phase flow capability See product details .
 Custom stacked design allows for space savings in parallel reactor systems. Available in 1/16″ HPLC and 1/8″ autoclave tube fittings.
Custom stacked design allows for space savings in parallel reactor systems. Available in 1/16″ HPLC and 1/8″ autoclave tube fittings.

Customer Case Study: Alternating Feeds to a Research Reactor. In cooperation with Oak Ridge National Lab’s Fuels, Engines and Emissions Research Center
 Fully automated catalysis reactor system by Integrated Lab Solutions (ILS) . ILS offers many different configurations, including a 32-Parallel Fischer-Tropsch fixed bed reactor system.
Fully automated catalysis reactor system by Integrated Lab Solutions (ILS) . ILS offers many different configurations, including a 32-Parallel Fischer-Tropsch fixed bed reactor system.
High Performance Mixed Phase Back Pressure Regulator
Application Focus: Hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen and other compounds and is used in industries such as food, pharmaceutical and petrochemical.
Hydrogenation is commonly performed in a fixed-bed catalytic reactor performed at elevated temperature and pressure to increase the density of the hydrogen and increase the rate of reaction. The control of reactor pressure is often critical, and can be difficult due to aggressive chemicals, high temperature and pressure, and the presence of mixed phase reactants.
The Equilibar® back pressure regulator excels in demanding applications such as reactor pressure control for hydrogenation.
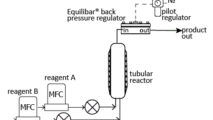
Applications Focus: Flow Chemistry
Continuous Flow Chemistry, or flow chemistry, is a popular way to run a process with advantages over batch chemistry. These advantages include more consistent quality, increased efficiency and easy scale-up from R&D to production. In flow chemistry, two or more reagents are mixed in a continuous manner through a reactor with flow, temperature and pressure controlled such that the desired chemical reaction can take place most efficiently.
Controlling pressure in flow chemistry is important for phase control, residence time, reaction speed and equilibrium management. An Equilibar BPR offers several advantages over other BPRs in flow chemistry applications. With its simple design, it controls over a wide flow range, offers superior precision, provides instantaneous control, and can handle extreme temperatures, corrosive chemistries as well as multi-phase flow found in flow chemistry.