Controlling pressure in flow chemistry is important for phase control, residence time, reaction speed and equilibrium management. For instance, reaction temperature is a key component in reaction efficiency but at high enough temperatures, a reagent can reach boiling point which negatively impacts the reaction. Controlling reaction pressure by adding a back pressure regulator to the outlet of the reactor can prevent reagent boiling.
Sometimes one of the reagents is a gas or sometimes an inert gas is introduced. By increasing the reactor pressure, the actual volume that the gas will occupy is lowered. This will leave more room for the liquid part of the reaction, thereby increasing the production rate of a reactor.
Additionally, in gas-liquid reactions, the pressure plays a role in the reaction speed. Often the gas must dissolve in the liquid before it can react. By increasing the pressure, the solubility of the gas is increased, which in turn increases the reaction speed.
When a chemical reaction is reversible, an equilibrium will form between reagents and product(s), slowing down reaction rates as equilibrium is reached. When a gas is involved in such a reaction, the equilibrium will be dependent on the pressure. At higher pressures, the equilibrium will shift towards the side of the reaction equation with the fewest gas molecules.
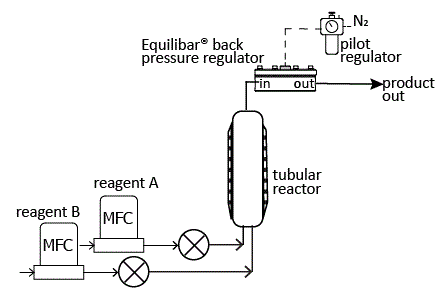
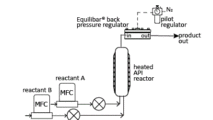
In the schematic above, an Equilibar® back pressure regulator (BPR) is installed at the exit of a tubular reactor for precise pressure control and process optimization. This schematic shows a high pressure reactor where the pilot regulator is controlling the dome of the Equilibar with Nitrogen gas. Other reactor designs may be lower pressure so that air may be used to pilot the dome-loaded Equilibar. A similar setup would also be effective when using a plug flow reactor (PFR).
Another type of continuous flow reactor is a continuously stirred tank reactor (CSTR) where reactants are continuously flowed into the CSTR, undergo a chemical reaction and exit at the same flow rate as the inlet stream to maintain a constant volume inside the reactor. Pressure control is important in this reactor design as well.
An Equilibar BPR has several advantages over other BPRs in flow chemistry applications. With its simple design, it controls over a wide flow range, offers superior precision, provides instantaneous control, and can handle extreme temperatures, corrosive chemistries as well as multi-phase flow found in flow chemistry. Because the supple diaphragm it its only moving part, the Equilibar back pressure valve is also easy to maintain on site in a few minutes.
Continuous flow chemistry is used in a variety of industries including petrochemical, specialty chemical, active pharmaceutical ingredients (API) and polymer chemistry.
Equilibar back pressure regulators are available in port sizes from 1/8” up with a variety of fitting types and pressure control ranges to meet the needs of your process design.
Contact an application engineer for more information about using an Equilibar back pressure regulator in your flow chemistry process.
 Equilibar stainless steel GSDH3 3/8” BPR for high pressure control up to 2500 psig (170 bar(g)). This one has Swagelok VCO fittings, with other fitting types available. Other models are available for lower pressure control and different port sizes.
Equilibar stainless steel GSDH3 3/8” BPR for high pressure control up to 2500 psig (170 bar(g)). This one has Swagelok VCO fittings, with other fitting types available. Other models are available for lower pressure control and different port sizes.
Download Brochure Request a Quote

Equilibar PTFE back pressure regulator used in continuous flow pharmaceutical research at MIT
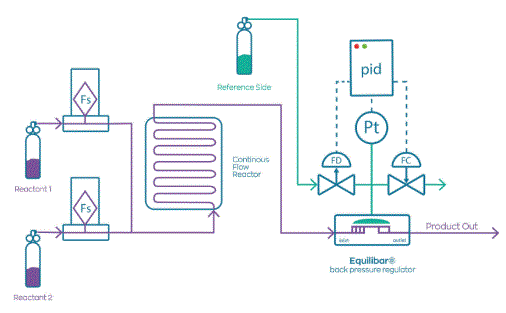
Pressure Control Solutions Blogs about all the reasons Equilibar back pressure valves are an excellent solution for flow chemistry, especially for his customer Flowid B.V. in The Netherlands.
Co-Authors:
Frans Arts, Owner at Eindhoven Process Technology – “Providing solutions in the realm of process technology to SMEs” Frans can be reached at frans@eindhovenprocesstechnology.nl
Diane Jacober, Marketing Specialist at Equilibar dianejacober@equilibar.com
Learn more about our back pressure regulator technology and how it works.
Watch this video how the Equilibar Back Pressure Regulator works.